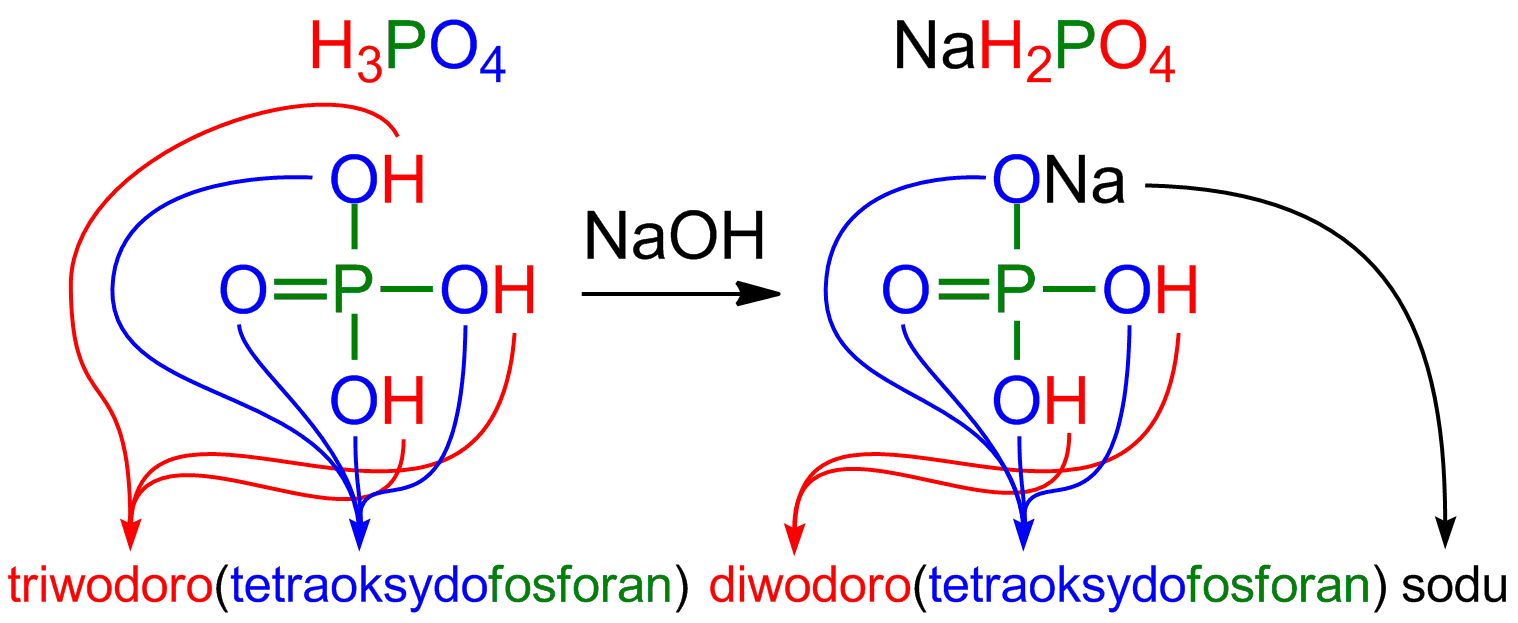
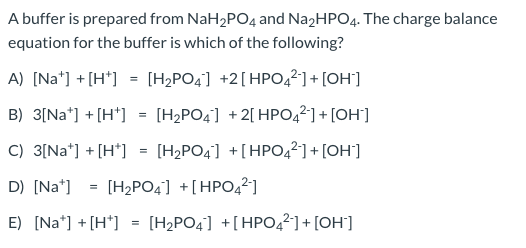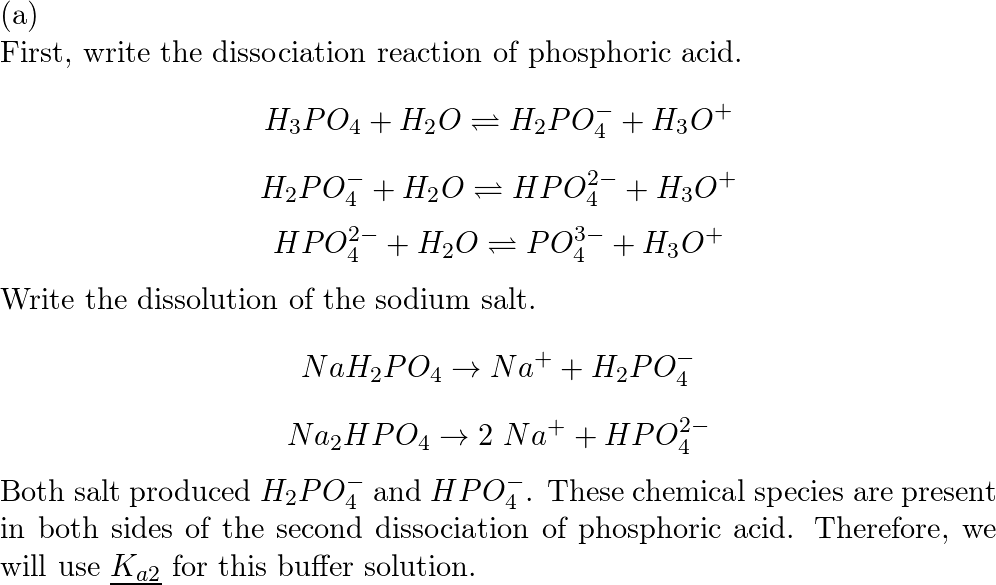The titration curve of fabrics treated with 6.0 % MA and 4.0 % NaH2PO4,... | Download Scientific Diagram
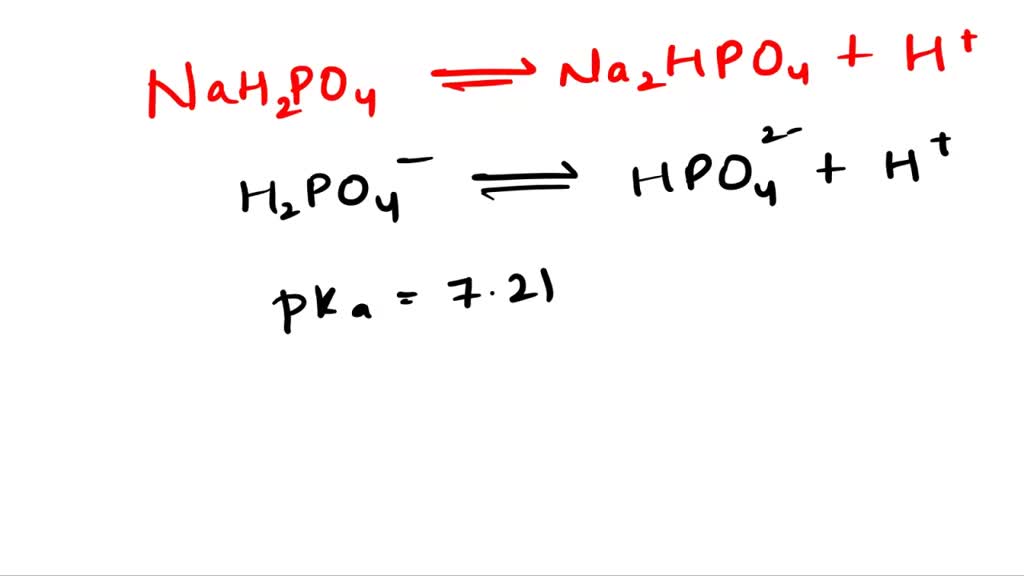
SOLVED: 1. Why is the equilibrium between the acid NaH2PO4, and its conjugate base Na2HPO4, a suitable buffer for maintaining intracellular pH (pH 6.9-7.3)?

SOLVED: Using the Henderson-Hasselbach equation solve the following: Prepare 150 mL of 0.5 M phosphate buffer; pH 8.2 Start with solid, anhydrous NaHzPO4 and NazHPO4 and water: For phosphoric acid; the proton

The equivalent weight of NaH2PO4 in the reaction NaH2PO4 + KOH ---- NaKHPO4 +H2O is 1) 158 2)60 3) - Chemistry - Some Basic Concepts of Chemistry - 13139619 | Meritnation.com

If 1,2,3,4 moles of H2PO4,NaH2PO4,Na2HPO4 and Na2PO4 respectively are mixed together to form an aqueous solution, the resulting pH is: Given values of Ka are: Ka1 = 10^-3 Ka2 = 10^-7 Ka3 = 10^-13

The equivalent weight of NaH2PO4 in the reaction NaH2PO4+KOH→NaKHPO4 + H2O (Given Atomic masses: Na = 23. K = 39, P = 31)

Monosodyum Fosfat (nah2po4) Gıda Susuz - Buy Msp/gıda Sınıfı,Msp/gıda Sınıfı/monoodium Fosfat Dihidrat,Monosodyum Fosfat Kimyasal Formülü/fiyatları Product on Alibaba.com

SOLVED: A buffer was made by mixing aqueous solutions of NaH2PO4 and Na2HPO4 together. This buffer is made by mixing two salts together. a. Write the balanced dissociation reaction for solid NaH2PO4
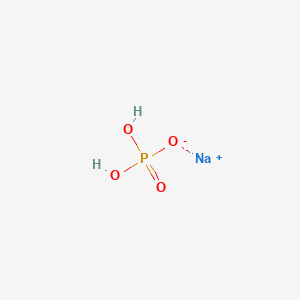
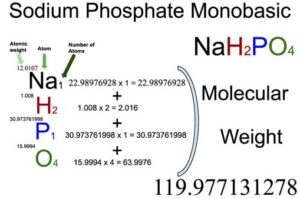
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $36.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $36.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%201KG.jpg)